
Special statement:
This article is for internal industry communication only and does not make any recommendations for specific brands or products.
The images presented in this article are only used to describe the facts and are not intended as advertisements for any products.
Minors are prohibited from accessing this article.
Product Highlights
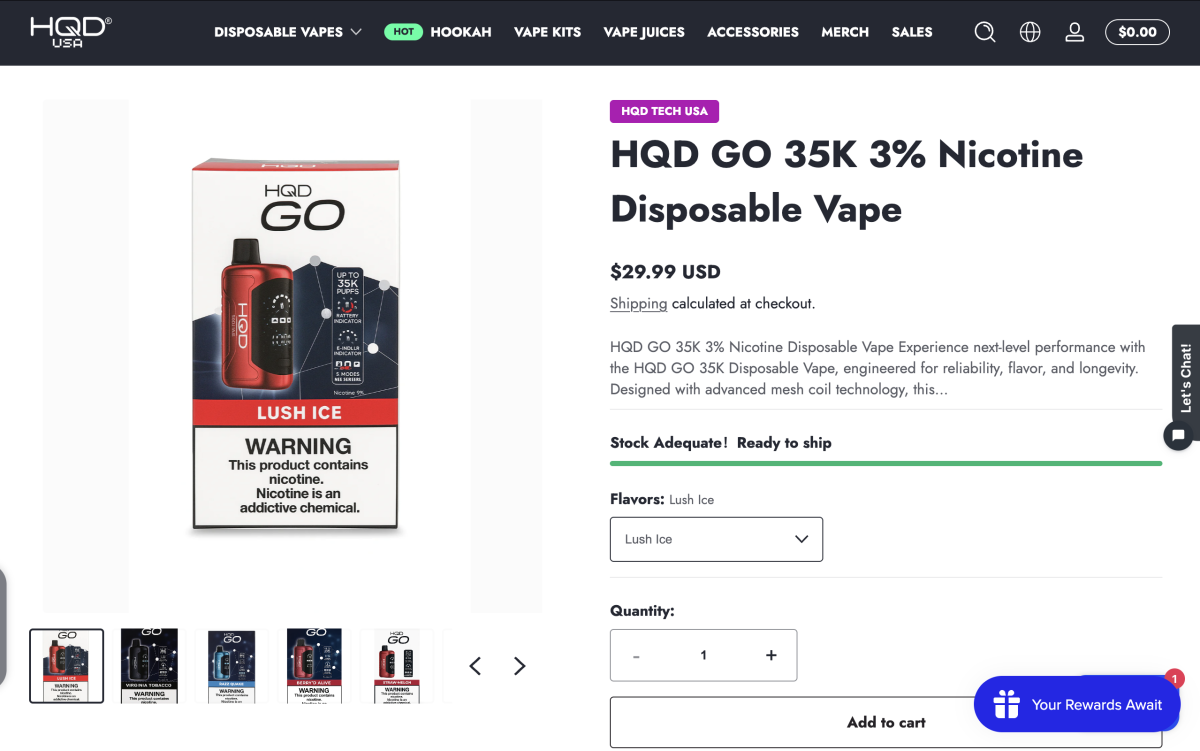
- Up to 35,000 puffs: Officially rated for up to 35K puffs; retail pages list an e-liquid capacity of 18 ml, equating to ~1,944 puffs per ml.
- Three power levels + adjustable airflow: Supports Soft / Regular / Boost modes with adjustable airflow.
- U.S. version nicotine 30 mg/ml: Lower than the common 5% in the U.S., positioned as a “low-nicotine, high-puff-count” combo.
- Price range & positioning (U.S.): Some retailers currently list it at about $29.99, which is on the higher end among high-puff disposables.
2Firsts, October 24, 2025 — HQD has launched the disposable e-cigarette HQD GO on its official website and across multiple U.S. retail channels. The model is officially rated for up to 35,000 puffs, offers Soft/Regular/Boost three-level power control and adjustable airflow. The U.S. version has a 30 mg/ml (3%) nicotine strength, and certain channels price it at $29.99.
30 mg/ml nicotine; up to 35K puffs (claimed)
2Firsts compiled the relevant specifications of the HQD GO, as summarized in the figure below:

From a specs standpoint, HQD GO is claimed to deliver up to 35,000 puffs. The vape retail site ohmcityvapes lists 18 ml of e-liquid, which converts to roughly 1,944 puffs per ml. For comparison, recent market releases VOZOL NEON PLUG MAX and SKE 30K Pro Max support about 1,500 puffs per ml.

The U.S. version of HQD GO has a 30 mg/ml nicotine concentration, lower than the 50 mg/ml commonly used in the U.S. market.
HQD’s official site showcases 6 device colors and 14 flavor options for HQD GO, spanning fruit, icy/cooling, chocolate, tobacco, and more.

Available on U.S. channels; priced at $29.99
HQD has listed the product on the HQD official site and U.S. e-cigarette retail websites. U.S. market retail pricing is around $29.99, as seen on sites such as HQDtechusa and Thevapersworld. In terms of pricing, HQD GO sits in the higher-priced tier across the e-cigarette market.
As of publication by 2Firsts, HQD GO has not received U.S. FDA marketing authorization via the PMTA pathway.
As a leading global NGP media and think tank, 2Firsts is dedicated to providing the latest product and technological information and insights to practitioners around the world in various categories such as e-cigarettes, heated tobacco products, and modern oral products. It aims to drive technological changes and innovations in NGP products worldwide, thereby offering tobacco consumers globally with safer products and lifestyles.
With a source of information covering the supply chains in China and global markets, 2Firsts product coverage has become one of the most influential platforms for new product and technology releases globally.
Contact 2Firsts for the following services:
1. Providing leads on new products and technologies;
2. Offering comments on products and technologies;
3. Seeking media coverage for your products;
4. Identifying sales channels for products.
Contact Information: Email: info@2firsts.com
Contact CEO Alan Zhao of 2Firsts on LinkedIn.
We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com









