
Special statement:
This article is for internal industry discussion only and does not make any recommendations for brands or products.
The images presented in this article are used solely to depict facts and do not serve as advertisements for any products.
This article is not intended for minors.
Shenzhen Kangertech Technology Co., Ltd., a manufacturer of e-cigarettes, recently launched a new type of e-cigarette called SUBOX PRO 50000 on its official website. According to the brand's promotion, this product is the "world's first disposable e-cigarette with triple control functions for nicotine, cooling sensation, and bubble/aroma.


Specifically, the product specifications of the Kanger SUBOX PRO 50000 are shown in the table below.
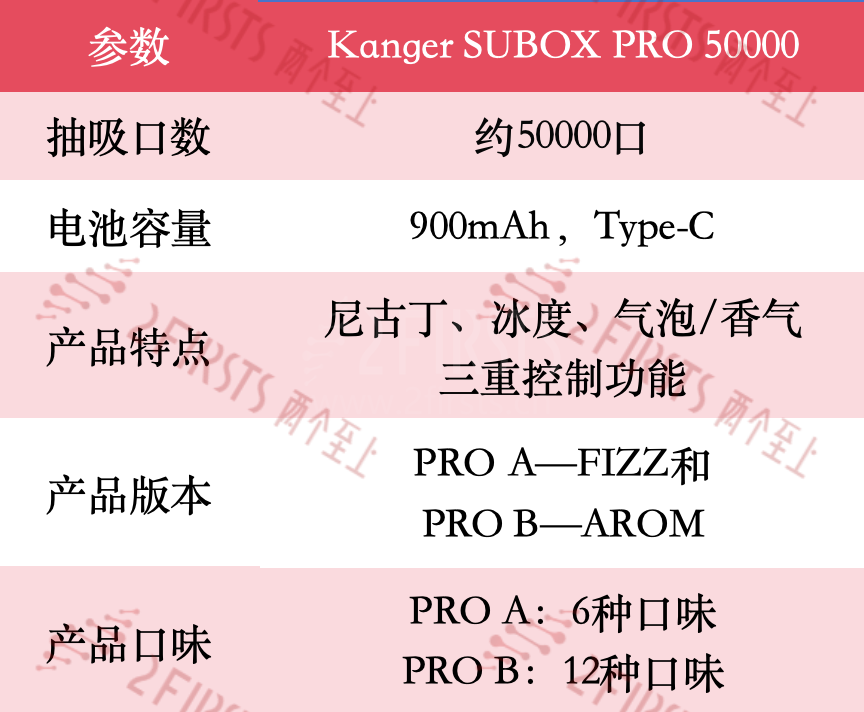
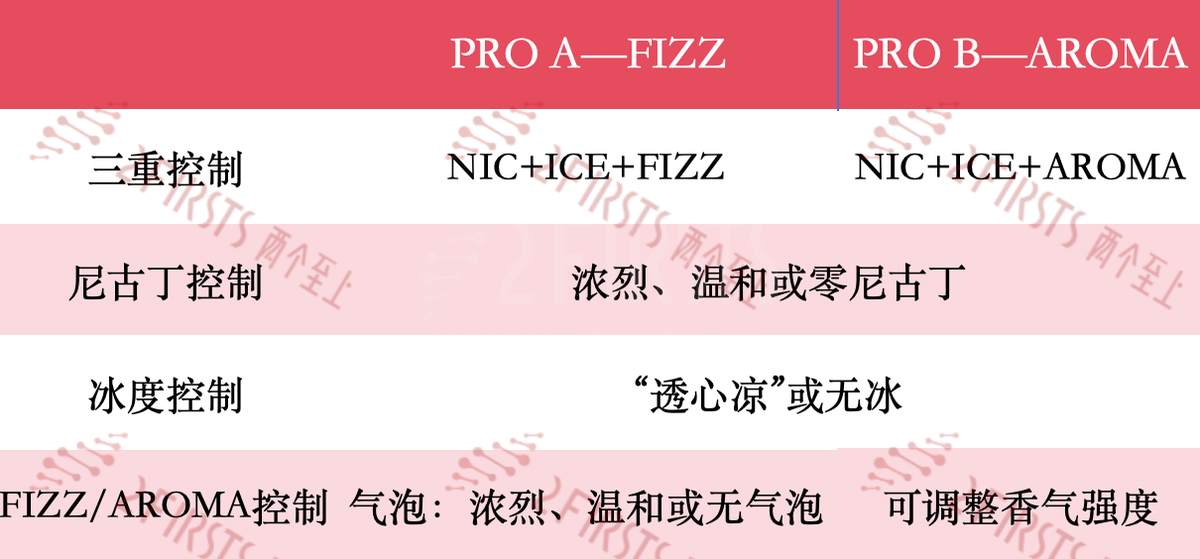
According to the e-cigarette product information website vapzvape.com, the SUBOX PRO 50000 is the "world's first disposable e-cigarette with triple control functions for nicotine, ice level, and bubble/scent." The product is available in two versions: PRO A - FIZZ and PRO B - AROMA. The PRO A version allows for adjusting nicotine concentration and ice level, as well as bubble strength control; while the PRO B version offers aroma control.

According to reports, KangerTech released the Kanger SUBOX 50000 in October, a product that features a large 50,000 puff count, a 40ml e-liquid capacity, and a low 0.35% nicotine concentration. (For more details, see "Product | KangerTech launches Kanger SUBOX 50000 with 50,000 puff count and 3D curved screen")
Compared to the previous Kanger SUBOX 50000, the new model maintains a consistent appearance but has improved the functionality of the screen. The SUBOX PRO 50000 has added fingerprint control, allowing users to make various settings and adjustments by simply touching the screen. Additionally, this product also has the capability to display real-time nicotine concentration, ice level, and bubble/aroma levels.
Currently, the new Kanger SUBOX PRO 50000 has been launched on the official KangerTech website and is being introduced on the e-cigarette product information website vapzvape. The product has not yet been released on other overseas e-cigarette distributors' websites.
In the global e-cigarette market, customizable flavor products are becoming a trend. Currently, adjustable flavor products on the market are mainly focused on single flavor adjustment brought by dual-tank products, as well as dual flavor adjustment provided by triple-tank products. The triple control function of the SUBOX PRO 50000 is groundbreaking in the current market.
2Firsts will continue to monitor the performance and market trends of e-cigarette products globally, providing the public with accurate and authoritative industry information.
2FIRSTS, a product column, is dedicated to providing readers with the latest information on new products in the tobacco industry. In order to gather and share first-hand information within the industry, 2FIRSTS welcomes readers to submit articles and share the latest products in the e-cigarette field.
If you have any unique insights or information, feel free to contact us anytime via email at info@2firsts.com.
We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com









