According to a report by the Manila Standard on June 4th, the Department of Trade and Industry (DTI)'s agency, the Bureau of Philippine Standards (DTI-BPS), has issued the first batch of Philippine Standards (PS) permits to manufacturers of heated tobacco products.
Deputy Secretary-General of the Consumer Protection Group of the Ministry of Commerce, Amanda Nograles, stated that
Starting from June 5, 2024, any e-cigarette products that do not carry the Product Standard (PS) mark, license, or ICC mark will be unable to enter the Philippine market.
The companies that have obtained PS licenses this time include Technocom System Sdn. Bhd. and Shenzhen Adwell Technology Co., Ltd. Both companies have successfully passed a rigorous review process and comply with the technical specifications of Republic Act No. 11900 of the Philippines, specifically the "Electronic Nicotine and Non-Nicotine Product Management Act.
According to the certification product list on the DTI-BPS website, the manufacturers of the IQOS ILUMA heated tobacco device, Venture Technocom System and Shenzhen Adwell Technology Co., Ltd., have both obtained licenses valid for three years, with a requirement to renew six months before expiration. It is worth noting that Venture Technocom System and Adwell Technology are the only two factories that have obtained PS logo approval for any products under the "e-cigarette law," with PMFTC Inc. and Royal Connections Inc. as authorized importers of IQOS ILUMA devices.
Furthermore, Noglares disclosed that in January 2025, the Ministry of Commerce will conduct random inspections of e-cigarette products without PS or ICC markings in the market. Sellers will have at least six months to deal with their inventory.
During a public lecture in October 2022, the DTI-BPS clearly stated that certification work is carried out to ensure that all products, equipment, and systems undergo rigorous analysis to meet quality standards and prevent illegal trade.
The Department of Trade and Industry (DTI) has announced that all heated tobacco and e-cigarette products will be required to obtain certification starting from June 5, 2024. Distributors and retailers can still sell uncertified electronic nicotine and non-nicotine products until January 5, 2025. After that date, only products with a valid PS mark or ICC label will be allowed for sale. The DTI has the authority to issue immediate recall, prohibition, or confiscation orders for businesses that do not comply with the regulations, to prevent their products from being sold or distributed in the public market.
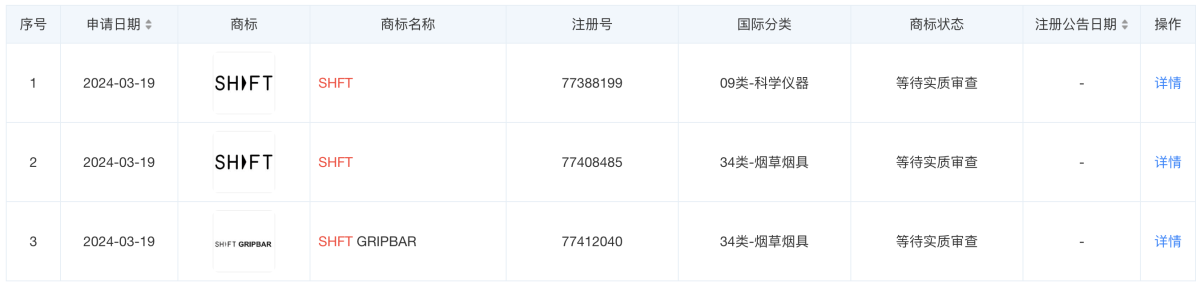
According to Tianyancha, Shenzhen Adiver Technology Co., Ltd. was established in 2019 and is located in Shenzhen, Guangdong Province. It is primarily engaged in the service industry. The legal representative is Zhang Zehong, with a registered capital of 1 million RMB. The business scope includes research and development, technical consultation, and sales of electronic atomization equipment, electronic atomizers and their components, and related equipment. In addition, records show that Adiver registered the SHFT trademark on March 19, 2024, and is currently awaiting substantive examination.
We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com