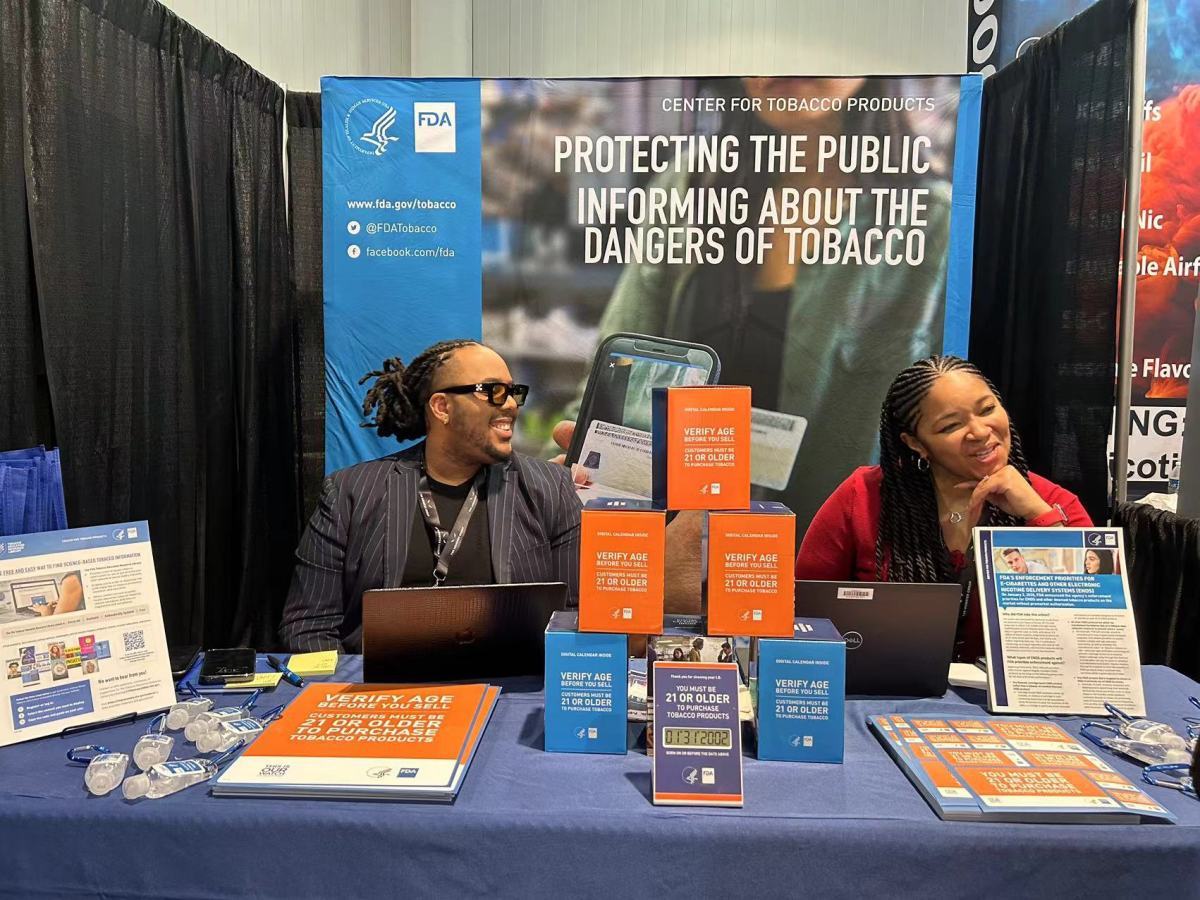
The largest tobacco exhibition in North America, TPE24, officially opened in Las Vegas on January 31st, Pacific Standard Time. The participation of the US e-cigarette regulatory agency, FDA, in this exhibition has sparked widespread attention and discussions within the e-cigarette industry. Industry insiders have been speculating on the motive behind FDA's participation.
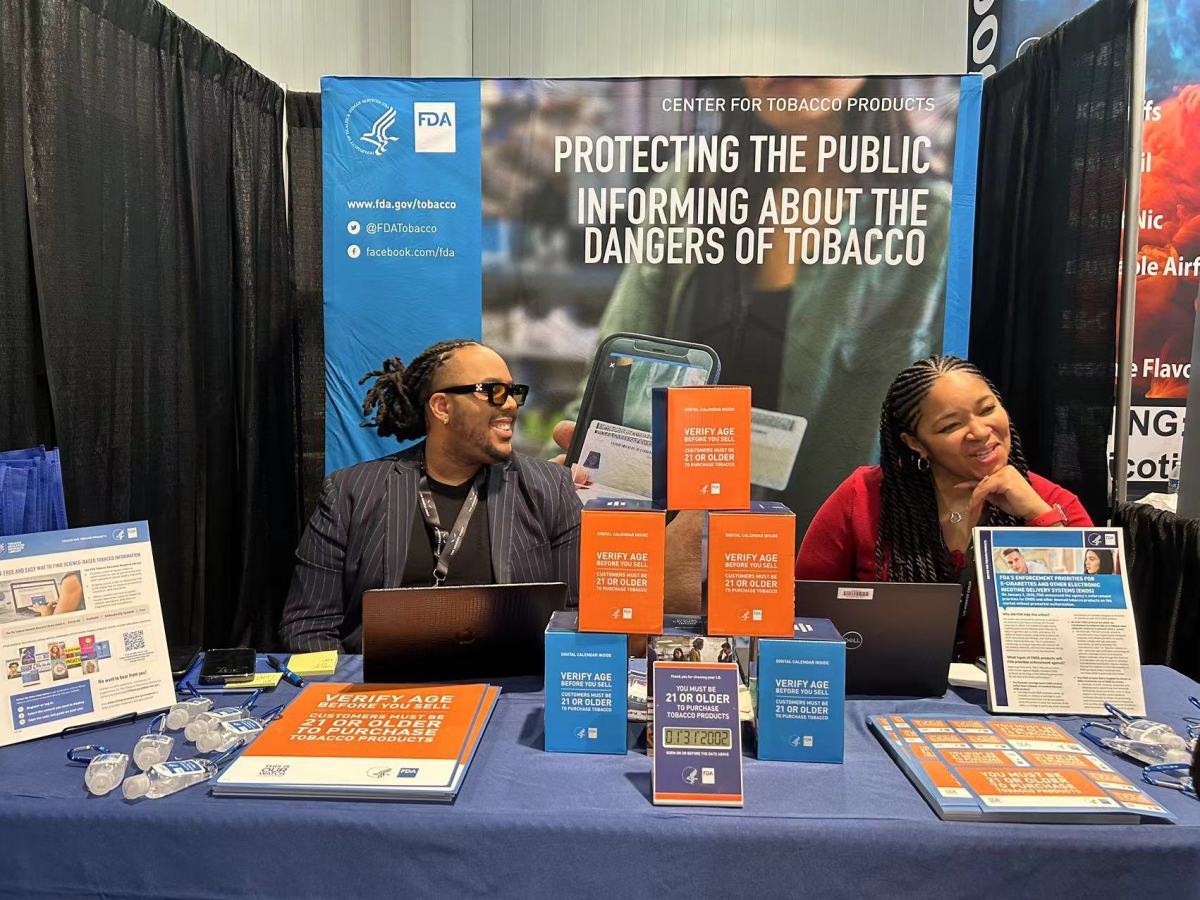
On the first day of the event, 2FIRSTS obtained information about the purpose of FDA's participation. It is understood that FDA's main objective of participating in the exhibition is to disseminate regulatory knowledge to e-cigarette companies, promote key policies related to FDA's regulation of e-cigarettes, and guide e-cigarette companies towards compliant sales practices. Additionally, they are providing free age verification devices to support e-cigarette companies in fulfilling their social responsibilities effectively.

In addition, 2FIRSTS also conducted interviews with FDA on-site personnel to discuss their future adjustment plans for PMTA. This content will be shared in detail during a live broadcast at the TPE exhibition and American market deep sharing session, held by 2FIRSTS on February 6. In this live event, 2FIRSTS Global Executive Editor Hou Yuhan and the US team will share the latest exhibition findings and delve into US product trends, policy directions, and more. For more information and registration details, please scan the QR code below to contact 2FIRSTS staff.


After the end of the exhibition, 2FIRSTS will be hosting a deep sharing event on TPE and the American market on February 6th. During this event, 2FIRSTS' Global Executive Editor-in-Chief, Yuna Hou, and the American team will share the latest discoveries from the exhibition and delve into topics such as product trends and policy direction in the United States. For more information and registration, please scan the QR code below to contact 2FIRSTS staff members.

We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com










