Special notice:
1.This article is for internal industry communication only and does not make any recommendations for brands or products.
2.The images displayed in this article are only used to describe facts and not intended as advertisements for any products.
3.This article is prohibited to be accessed by minors.
Key Points:
·Multiple brands announce new product launches simultaneously, including mainstream brands such as ELFBAR, SKE, and AL FAKHER. The new SKUs cover rechargeable devices, pre-packaged e-liquids, and open-ended kits.
·Taste Differences: AL FAKHER and Starbuzz announce multiple 6mg low concentration fruit/sweet flavor SKUs.
·Open Equipment Display: Brands such as INNOKIN, VOZOL, and VOOPOO have launched multiple refillable open devices.
【By 2Firsts】2FIRSTS queries of the UK Medicines and Healthcare products Regulatory Agency (MHRA) e-cigarette product notification database revealed that brands such as SKE, JNR, AL FAKHER, VOZOL, and INNOKIN all had new SKUs announced in July. These new SKUs, once approved by the MHRA, indicate that they have received marketing authorization and will soon be officially entering the UK market for sale.
2Firsts has compiled and organized the information from the Medicines and Healthcare products Regulatory Agency's (MHRA) e-cigarette product notification database since July. This includes new SKUs for various types of e-cigarette brands. Below are the details of the notifications.
ELFBAR and SKE have Each Released Public Announcements for Two SKUs
In the "ELECTRONIC CIGARETTE - RECHARGEABLE, DEVICE ONLY ANY RECHARGEABLE WHICH CAN ALSO BE USED AS A REFILLABLE SHOULD BE REPORTED UNDER THE REFILLABLE CATEGORY" product category, a total of 20 SKUs have been updated as of July 9th.
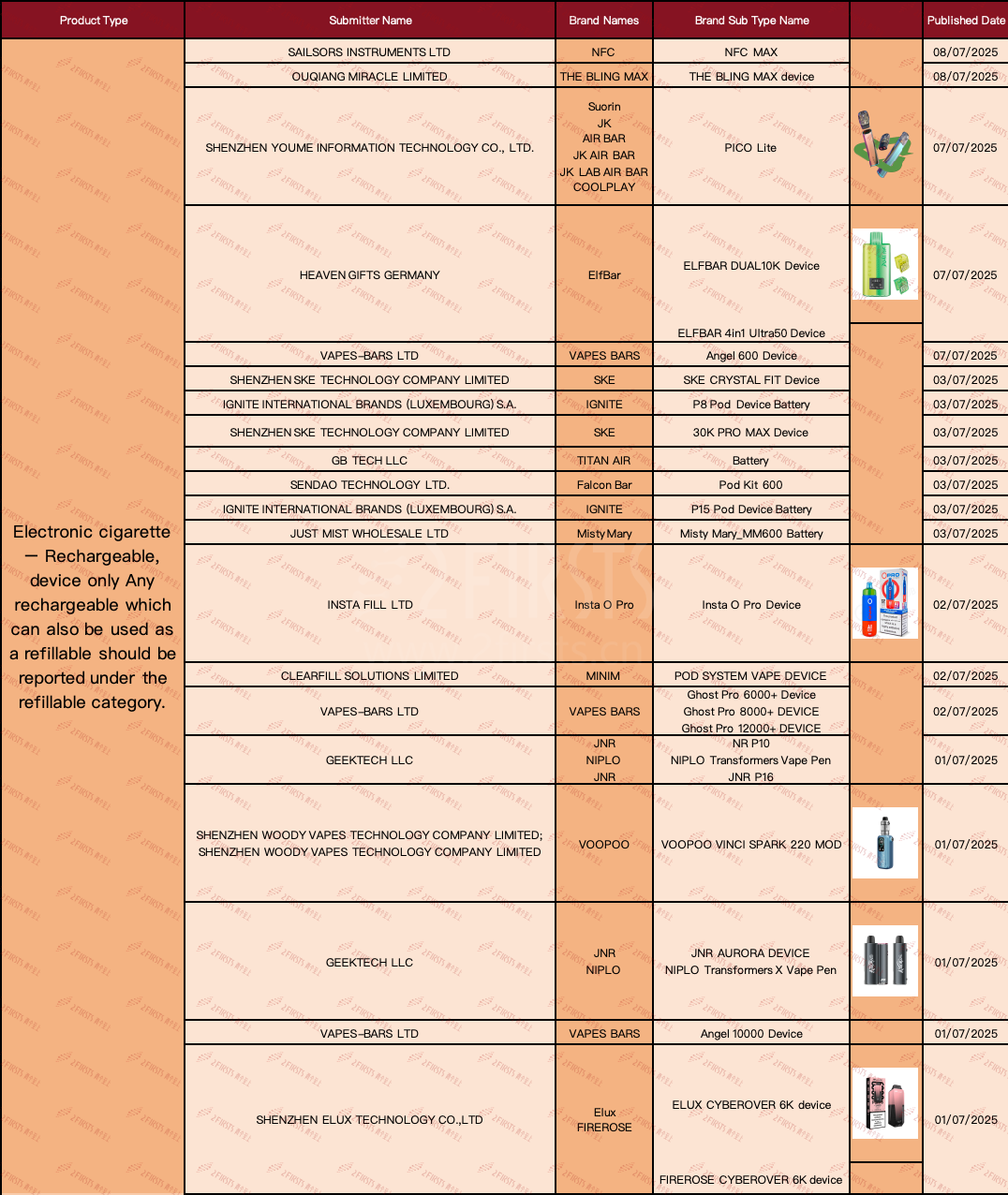
·ELFBAR brand officially announces the launch of two new devices: the ELFBAR DUAL10K Device and the ELFBAR 4in1 Ultra50 Device.
·SKE brand announces the SKE CRYSTAL FIT Device and the 30K PRO MAX Device.
·IGNITE brand announces the release of the P8 Pod Device Battery.
·VOOPOO brand announces the release of the VOOPOO VINCI SPARK 220 MOD.
·ELUX and FIREROSE have announced the ELUX CYBEROVER 6K device and FIREROSE CYBEROVER 6K device, respectively.
Al Fakher and Starbuzz have Announced Multiple 6mg Specification SKUs
In the product category "ELECTRONIC CIGARETTE – REFILLABLE, PLACED ON THE MARKET WITH ONE TYPE OF E-LIQUID (FIXED COMBINATION)", a total of 23 SKUs have been updated as of July 9th.
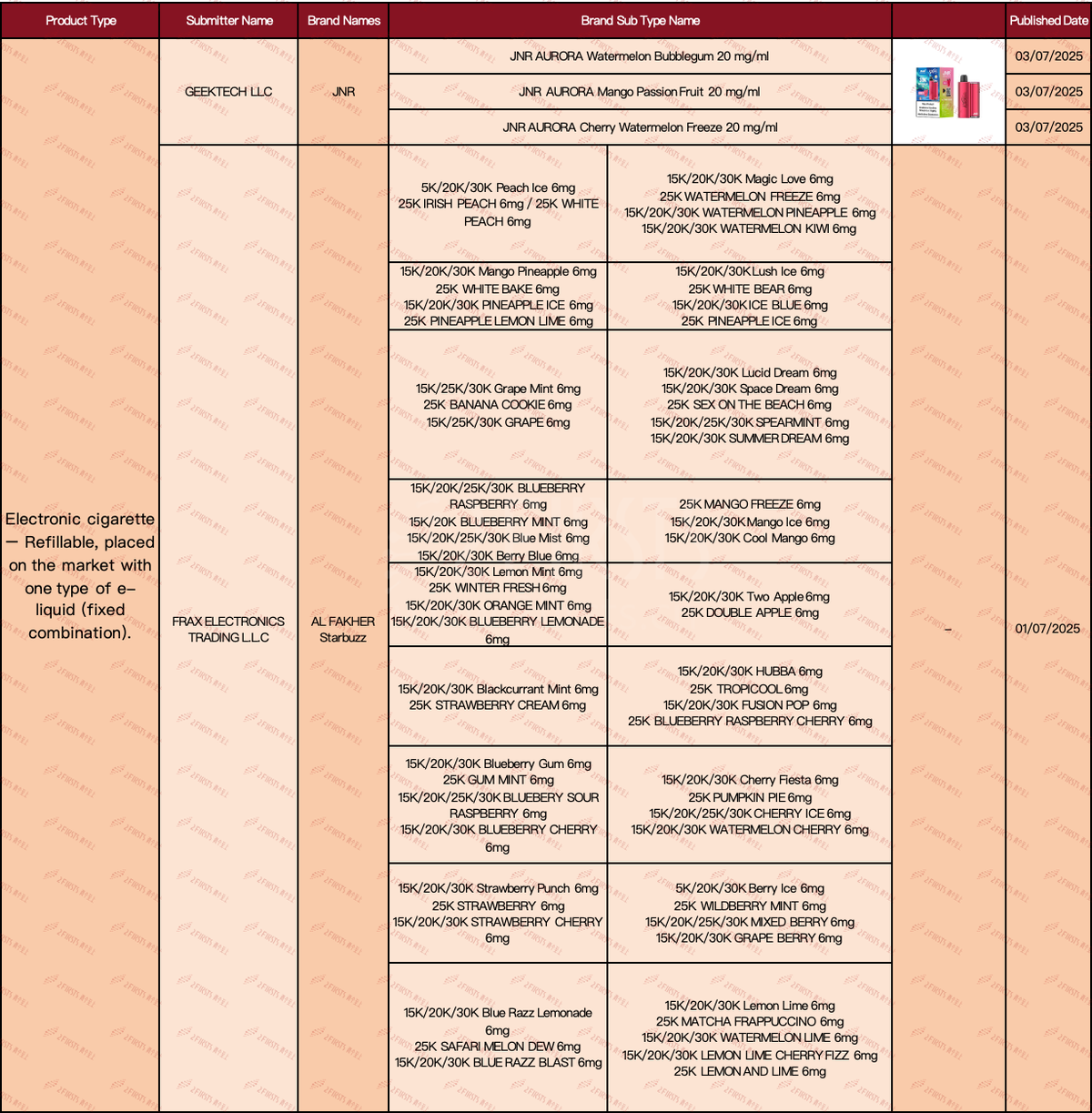
·The nicotine concentration of JNR brand's new SKUs is 20mg/ml, which is the maximum concentration allowed for e-liquid under UK/EU TPD/TRPR regulations.
·The brands Al Fakher and Starbuzz (submitted by PRANK ELECTRONICS TRADING L.L.C) unveiled multiple flavor SKUs in July.
·The nicotine concentration of the new SKUs from the AL FAKHER and Starbuzz brands is 6mg.
·The new SKUs of AL FAKHER and Starbuzz brands have a draw count ranging from 5,000 to 30,000.
·The new SKUs from AL FAKHER and Starbuzz brands focus on fruit/sweet flavors.
Innokin, Vozol, and Voopoo Announce New Open Pod Systems
In the "ELECTRONIC CIGARETTE - REFILLABLE, DEVICE ONLY" product category, a total of 15 SKUs have been updated as of July 9th.
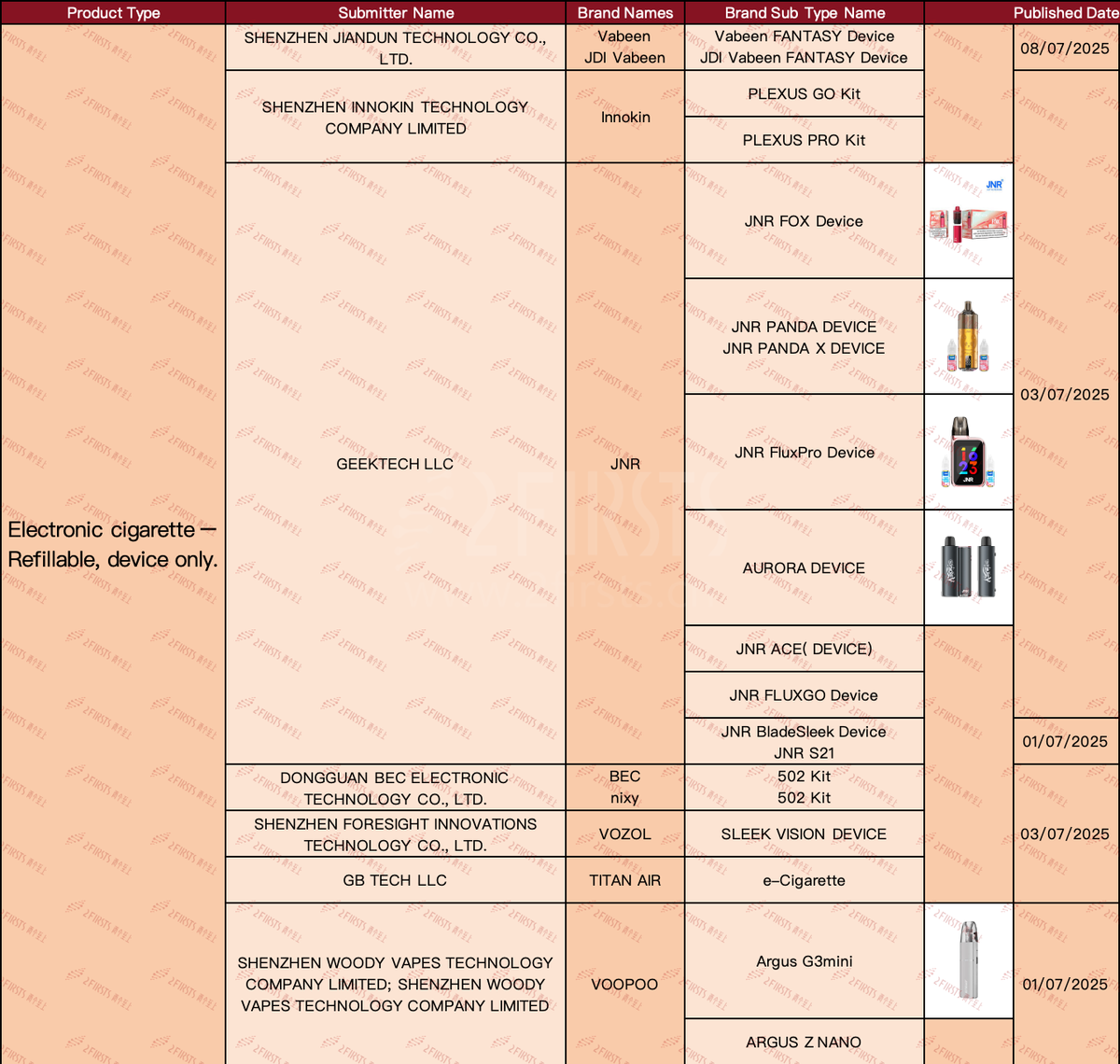
·Innokin brand announces PLEXUS GO Kit and PLEXUS PRO Kit.
·JNR announces new products including the JNR FOX Device, JNR PANDA DEVICE, JNR PANDA X DEVICE, JNR FluxPro Device, AURORA DEVICE, JNR ACE( DEVICE), JNR FLUXGO Device, JNR BladeSleek Device, and JNR S21.
·VOZOL brand announces SLEEK VISION DEVICE.
·The VOOPOO brand has unveiled the Argus G3mini and ARGUS Z NANO models.
In the product category "Kit - Pack containing more than one different e-cigarette device and/or more than one different refill container/cartridge," there have been a total of 7 SKUs updated as of July 9th.
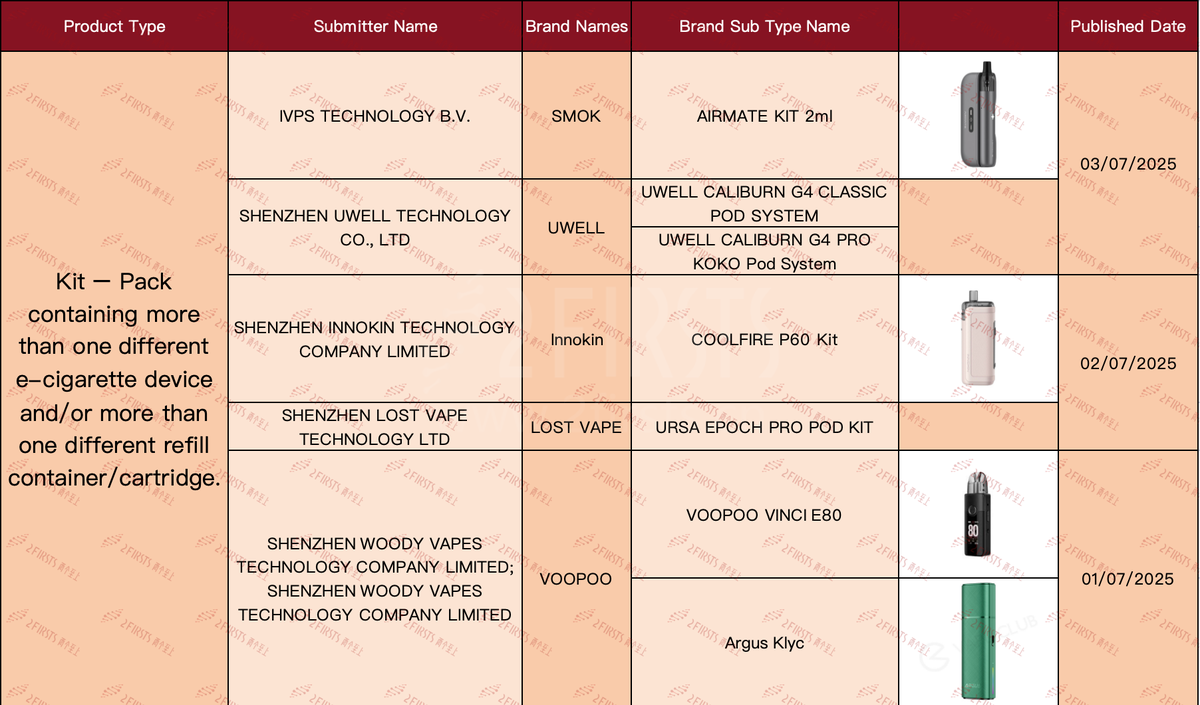
Involving brands such as SMOK, UWELL, INNOKIN, LOST VAPE, and VOOPOO.
On June 1st, the UK officially implemented a nationwide ban on the sale of disposable e-cigarettes, prohibiting the sale of products that cannot be refilled with e-liquid or have non-rechargeable batteries.
As a leading global media and think tank in NGP, 2Firsts is committed to providing the latest product and technology information and insights for practitioners worldwide in various categories such as e-cigarettes, heated tobacco products, and modern oral products. This aims to drive technological change and innovation in NGP products globally, ultimately bringing safer products and lifestyles to tobacco consumers worldwide.
With its information sources covering the supply chain in China and global markets, 2Firsts' product coverage has become one of the most influential platforms for the introduction of new products and technologies worldwide.
Welcome to contact 2Firsts for:
1. Providing leads on new products and technologies;
2. Providing comments on products and technologies;
3. Seeking media coverage for your products;
4. Collecting sales channels for products;
...
Contact information:
1. Email: info@2firsts.com
2.Contact 2Firsts CEO Alan Zhao on LinkedIn.
We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com