
Special Statement:
1.This article is intended for internal industry communication only and does not make any recommendations for specific brands or products.
2.This article is prohibited for minors to access.
3.The images presented in this article are used solely to depict facts and are not intended as advertisements for any products.
Product highlights:
·The 1.47 inch HD touchscreen interaction: HQD APEX can display real-time battery power, wattage, and work status.
·Wide range power output of 5-45W: HQD APEX is compatible with 0.4Ω, 0.8Ω, 1.2Ω three-level replaceable pods, supporting MTL (mouth to lung) to DTL (direct to lung) mode switching.
·Wide range power output: Compared to similar products (such as VOZOL ACE PRO, Geekvape Soul 2 Pod Kit), HQD APEX has the widest power range (5W-45W).
It is worth noting that e-cigarette brand HQD has launched a new open system - APEX on its official website, featuring a 1.47 inch HD touchscreen and supporting 5-45W wide range power output. At present, it has not yet been listed on e-cigarette distributor websites.
Equipped with a 1,000mAh battery for wide-range power output of 5-45W
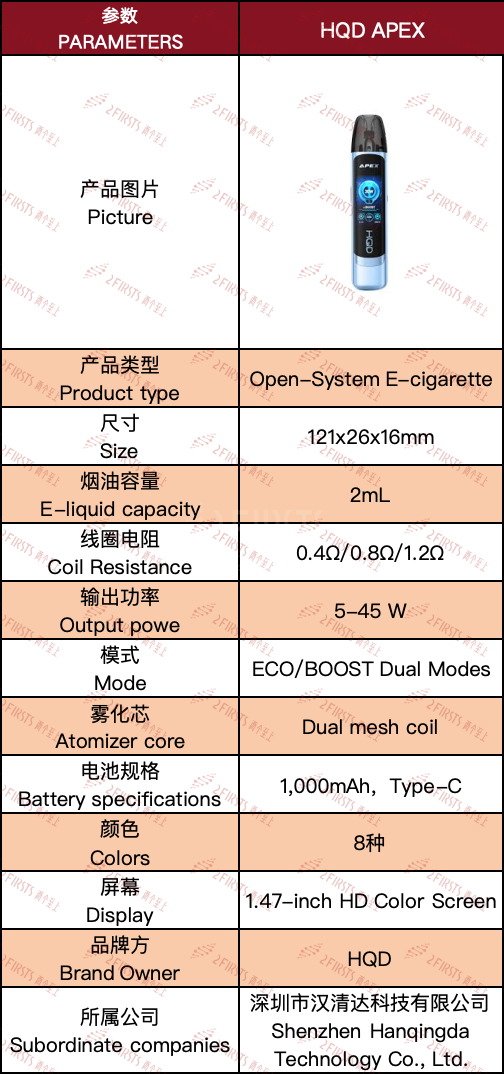
2Firsts collected the parameter information of HQD APEX, as shown in the table below:
Prior to this, the e-cigarette brand VAPORESSO had also released an open system named "APEX" - the Vaporesso Apex Pod Kit. It is reported that a tactical competitive game called "Apex Legends" launched in 2019 also uses this name.

From a product specification point of view, the HQD APEX is equipped with a 1,000mAh battery, which is lower compared to similar products such as the VOZOL ACE PRO (1400mAh) and Vaporesso Apex Pod Kit (2000mAh).
In addition, the HQD APEX is equipped with three different resistance replaceable pods of 0.4Ω, 0.8Ω, and 1.2Ω, corresponding to an output power range of 5W to 45W. Compared to several other brands such as VOZOL, Geekvape, FRESOR, the HQD APEX has a wider power output range, supporting the switch from MTL (mouth-to-lung) to DTL (direct-to-lung) modes.

It is worth noting that, while the HQD APEX product page displays three different resistance values of replaceable pods (0.4Ω, 0.8Ω, 1.2Ω), the official website states that the product is currently available in 0.8Ω and 1.2Ω variants. However, HQD APEX plans to release a version compatible with 0.4Ω pods in the future.

Equipped with a 1.47-inch HD touchscreen
Like many other multi-brand cartridge-style, open-system products that have been introduced before, the HQD APEX is equipped with a 1.47-inch HD touchscreen that can display real-time information such as battery life, power output, and operational status, and supports animated feedback. The product also features airflow adjustment on the side, and includes a side button that provides access to all functions.

Previously, the VOZOL brand launched a pod system e-cigarette product called the VOZOL Ace Max, which also features a 1.47-inch touchscreen display showing data such as power, puff count, and battery level.

Online sales have not been conducted yet
Currently, HQD APEX has been launched on the HQD brand official website and is being promoted simultaneously on the brand's Facebook and Instagram official accounts. The HQD brand has now released a variety of open-system products including HQD ZEST, HQD CIRAK, and HQD EOS 2.0.
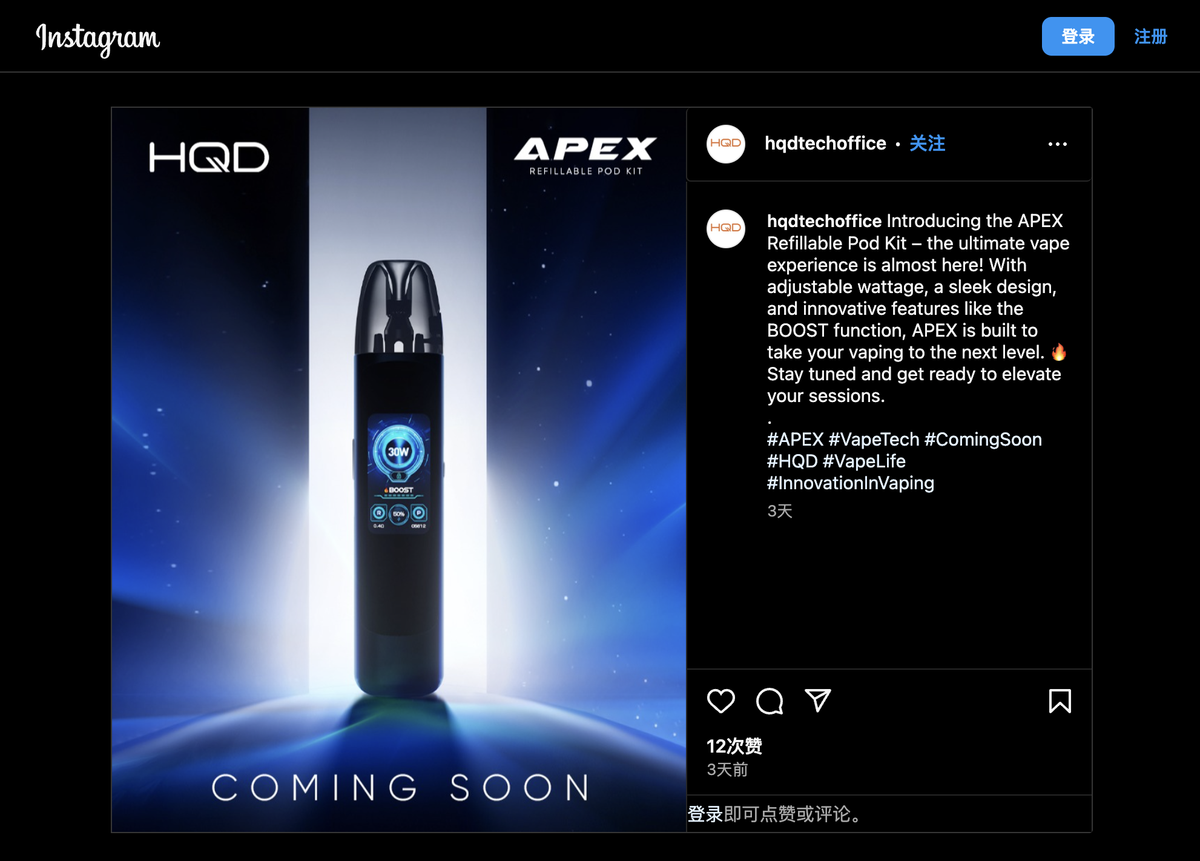
HQD APEX is currently not available for sale on overseas e-cigarette distributor websites; a search on the UK Medicines and Healthcare products Regulatory Agency (MHRA) e-cigarette product announcement database did not yield any information on HQD APEX, suggesting the product may not be targeted towards the UK market.
2FIRSTS focuses on providing readers with the latest information on new products in the tobacco industry. We welcome contributions from readers to help us gather and share firsthand information in the e-cigarette field.
If you have any unique insights or information, please feel free to contact us anytime via email at info@2firsts.com, or connect with 2Firsts CEO Alan Zhao on LinkedIn.
We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com










