
The Independent British Vape Trade Association (IBVTA) successfully hosted its annual forum on 22 October at the Birmingham National Conference Centre.
The forum brought together policy makers, regulators, law enforcement, industry experts and market practitioners from the UK e-cigarette industry to discuss innovative challenges, future regulatory trends, business insights, tax issues and more. 2FIRSTS attended the conference and provided live coverage of the event.

During the forum, Marsha Bell, representing the UK's Department for Trade Standards, pointed out that local authorities with limited resources face significant challenges in dealing with the rapid growth of the e-cigarette market. She mentioned that law enforcement agencies are struggling with how to store and dispose of the large quantities of non-compliant products they seize.
As the demand for illegal e-cigarettes continues to grow, the number of products seized is increasing and local authorities often lack the space and resources to deal with these items properly.

Craig Copland, MHRA E-cigarette Notification Manager, emphasised in a group discussion the importance of a thorough understanding of the 2ml e-liquid limit in the market. He believes that innovative products such as 2+10 and 4in1 reflect progress in the market and is positive about the nature of these products.

IBVTA member Liam Humberstone's presentation focused on 'Product innovation and how manufacturers should respond'. He highlighted the rise of disposable products such as ELFBAR, LOSTMARY and SKE, stating that the market needs more sustainable products to meet consumer demand.
Humberstone referred to large quantities of smuggled products as "opportunists", noting that these products violate UK regulatory policy and impact on compliant markets.

In addition, local British consultancy NIQ presented a recent research report on the e-cigarette market at the forum, which revealed the most popular e-cigarette brands on the current market, including ELFBAR, LOSTMARY, VUSE, SKE, IVG and GOLDBAR. Among them, the most popular brand is SKE and the most popular flavour is pineapple.

In terms of tax policy, UK-based consultancy Counterfactual also provided detailed insights into the current tax status and potential future trends of the e-cigarette industry.
In addition, participating organisations such as DEFRA, Wastecare and GAP shared their efforts and achievements in recycling e-cigarette products and protecting the environment, emphasising the industry's responsibility and commitment to sustainable development.
These discussions not only provided participants with valuable market and policy information, but also offered new ideas and solutions for environmental practices within the e-cigarette industry.
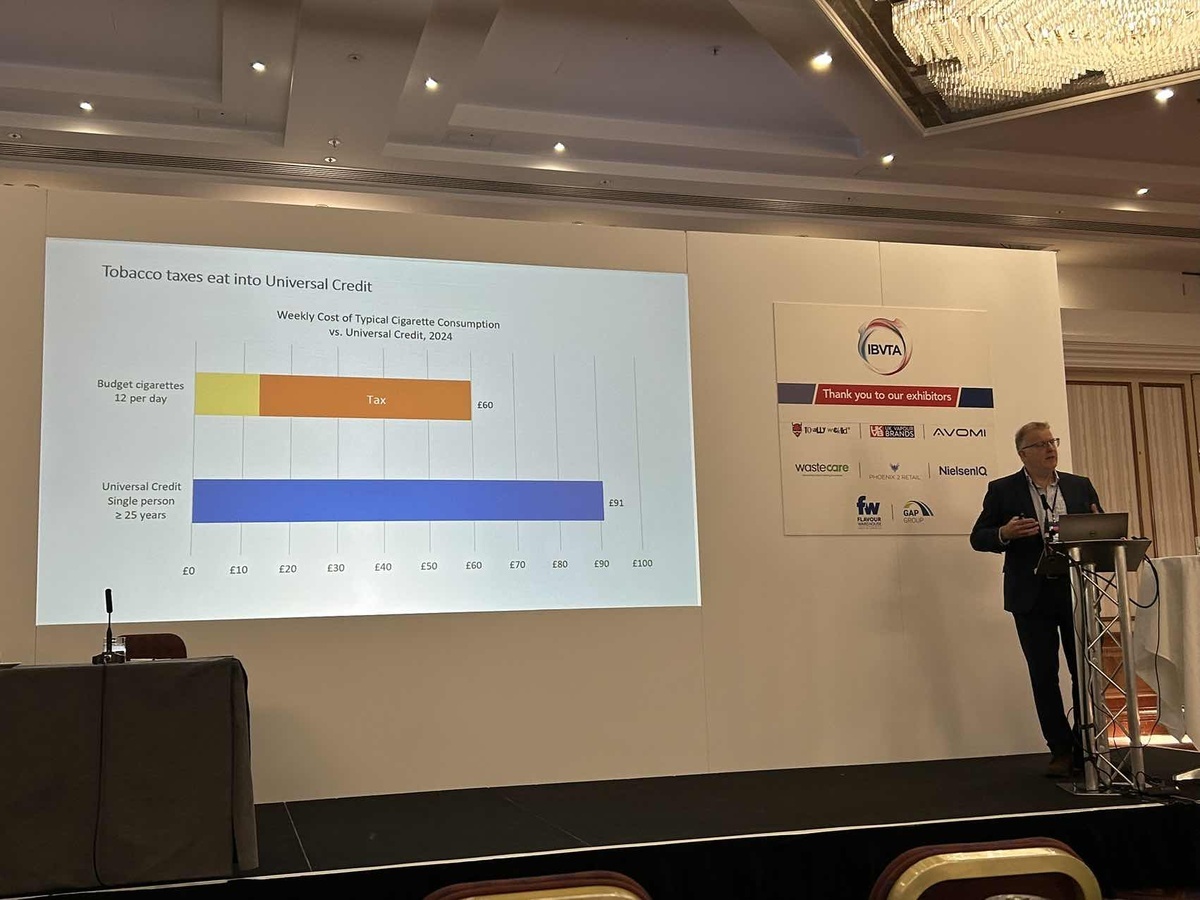
Finally, Marcus Saxton, Chairman of IBVTA, gave a detailed overview of the association's support for the All-Party Parliamentary Group (APPG) in the UK Parliament. He mentioned that the APPG is committed to providing a platform for discussion within Parliament on the issue of e-cigarettes, with a particular focus on appropriate regulation and industry responsibility.

We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com










