
Special announcement:
This article is intended for internal industry research and communication purposes only, and does not make any brand or product recommendations. Minors are prohibited from accessing it.
In the UK market, the camp of disposable e-cigarettes that are TPD compliant with over 5000 puffs has welcomed a new member.
The e-cigarette retail website VAPEUK recently launched a disposable product called FEOBA Big Bar 6000. This disposable e-cigarette enters the market with a capacity of 6000 puffs, priced at £12.99, which is comparable to another 6000 puff product, LOST MARY BM6000, also available on VAPEUK.
The design of FEOBA Big Bar 6000 also follows the common configuration of large-capacity products, using a combination design of "2ml+10ml".

According to the information provided, the FEOBA Big Bar 6000 features a transparent oil reservoir and a cotton-free design, with a built-in 1750mAh battery that claims to provide a user experience of 6000 puffs. The FEOBA Big Bar 6000 introduces the "PrecisionFlow™ HBS (Hydraulic Balance System)", which is said to be specifically designed for this Mesh platform to control the dosage of electronic liquid and adjust the air pressure for each inhalation.
The UK Medicines and Healthcare products Regulatory Agency (MHRA) e-cigarette search system shows that the product was announced on June 12th as an "e-cigarette- refillable, market released in a fixed combination with an electronic liquid.
The company in question is FEOBA Technology LTD, which is registered in London, England. According to the news section on FEOBA's website, the company participated in last year's Vaper Expo at the NEC in Birmingham, UK, but there is no further information available. The official website states that FEOBA was established in 2018, with headquarters in Europe, a design center in Marseille, France, and a joint venture with one of China's top 3 groups, boasting strong research and development, technology, and manufacturing capabilities.

According to information from the UK government's business inquiry platform, FEOBA Technology LTD was established on January 16, 2023, and has been operating for 1 year and 5 months. The latest submitted confirmation statement shows that the company currently has 1 active director and 1 active secretary, both being MENG, Qinghui.

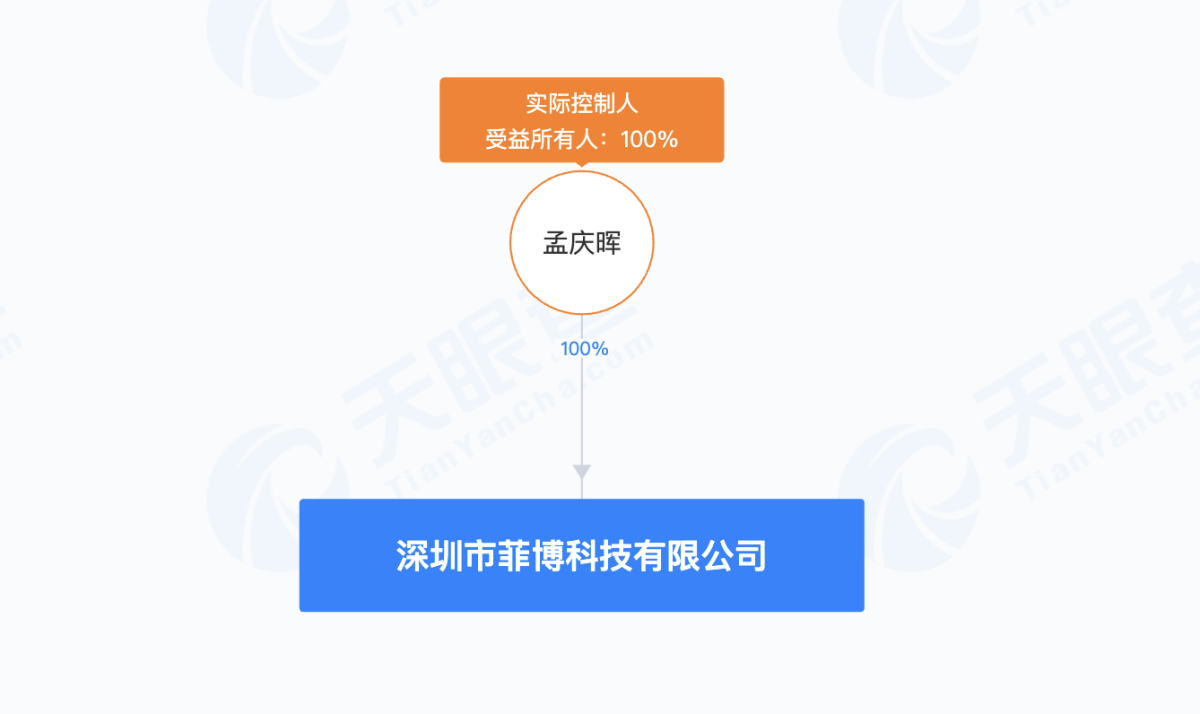
According to a search on Tianyancha, there is a connection between FEOBA Technology LTD and Shenzhen Feibo Technology Co., Ltd., with the latter's actual controller being Meng Qinghui. However, a search on Ali Trademark shows that FEOBA's trademark in Class 34 for tobacco and smoking accessories was applied for by New Zealand (Shenzhen) Technology Co., Ltd. and is currently pending approval, with no intellectual property owned by Shenzhen Feibo Technology Co., Ltd.

Noteworthy is that VAPEUK has several disposable e-cigarette products that comply with TPD regulations. However, the current maximum number of puffs remains at 6000, with most products staying within the range of 5000 puffs.
We welcome news tips, article submissions, interview requests, or comments on this piece.
Please contact us at info@2firsts.com, or reach out to Alan Zhao, CEO of 2Firsts, on LinkedIn
Notice
1. This article is intended solely for professional research purposes related to industry, technology, and policy. Any references to brands or products are made purely for objective description and do not constitute any form of endorsement, recommendation, or promotion by 2Firsts.
2. The use of nicotine-containing products — including, but not limited to, cigarettes, e-cigarettes, nicotine pouchand heated tobacco products — carries significant health risks. Users are responsible for complying with all applicable laws and regulations in their respective jurisdictions.
3. This article is not intended to serve as the basis for any investment decisions or financial advice. 2Firsts assumes no direct or indirect liability for any inaccuracies or errors in the content.
4. Access to this article is strictly prohibited for individuals below the legal age in their jurisdiction.
Copyright
This article is either an original work created by 2Firsts or a reproduction from third-party sources with proper attribution. All copyrights and usage rights belong to 2Firsts or the original content provider. Unauthorized reproduction, distribution, or any other form of unauthorized use by any individual or organization is strictly prohibited. Violators will be held legally accountable.
For copyright-related inquiries, please contact: info@2firsts.com
AI Assistance Disclaimer
This article may have been enhanced using AI tools to improve translation and editorial efficiency. However, due to technical limitations, inaccuracies may occur. Readers are encouraged to refer to the cited sources for the most accurate information.
We welcome any corrections or feedback. Please contact us at: info@2firsts.com










